Advancing surgical care for gastroparesis
Newly expanded coverage
As of January 1, 2026, Gastric Electrical Stimultion (GES) with Enterra Therapy is now covered1 in the Amublatory Surgical Center (ASC) setting by Medicare for patients with diabetic or idiopathic gastroparesis experiencing chronic nausea and vomiting who have failed medical therapy.
According to the Humanitarian Use Device (HUD) approval granted by the FDA, Enterra Therapy is indicated for the treatment of chronic, drug refractory nausea and vomiting associated with gastroparesis caused by diabetes or unknown cause (idiopathic) gastroparesis in patients aged 18 to 70 years.

Why consider Enterra Therapy?
Enterra Therapy offers a clinically proven,2-5 minimally invasive therapy addressing an unmet patient need. Surgeons can now offer an established option for patients with diabetic or idiopathic gastroparesis who remain symptomatic despite having tried other treatment options.
Familiar, minimally invasive procedure
Designed to fit into standard surgical workflows:
- Typically performed laparoscopically or robotically
- Average procedure time: 60–90 minutes under general anesthesia
- Most patients are discharged the same day
- Well-suited to leverage existing surgical skills, equipment, and infrastructure
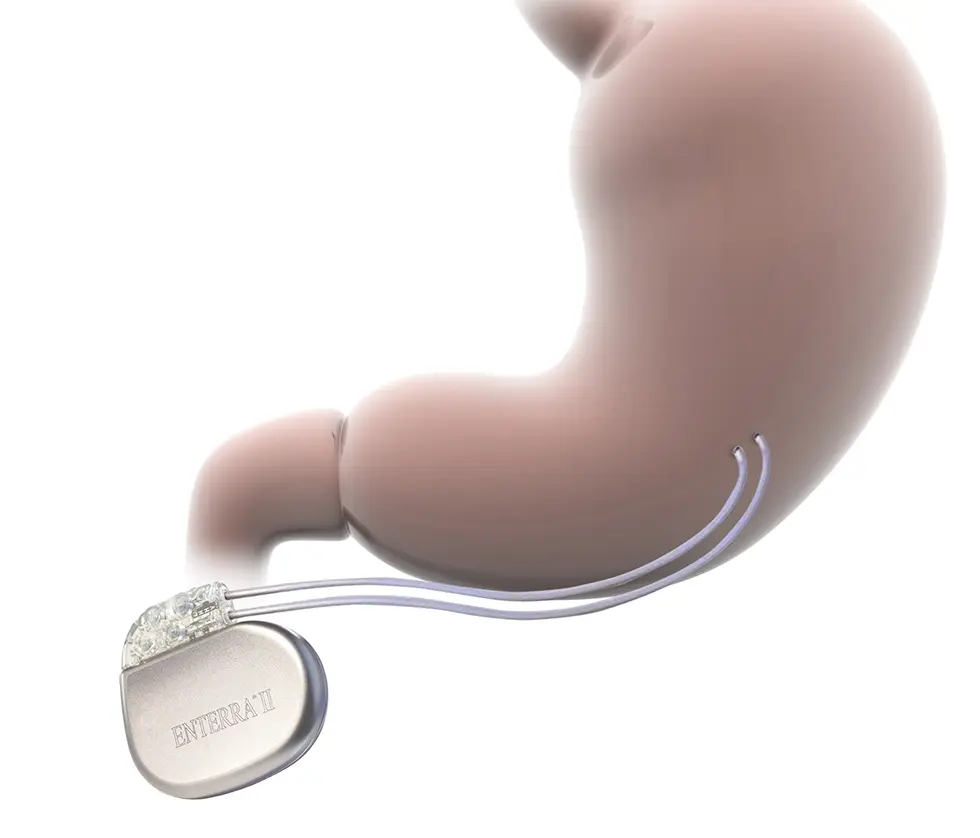
Procedure overview
Electrodes are implanted in the stomach wall
Enterra Therapy implant procedure typically takes 60-90 minutes in a minimally-invasive laparoscopic or robotic surgical procedure, where the leads are placed on the serosal surface of the stomach’s greater curvature.
Neurostimulator is implanted in subcutaneous abdominal pocket
The neurostimulator is placed in subcutaneous tissue, typically in the abdomen.
Therapy is adjusted non-invasively
Post-procedure, Enterra Therapy can be adjusted for symptom control via a programmer in an outpatient clinic.
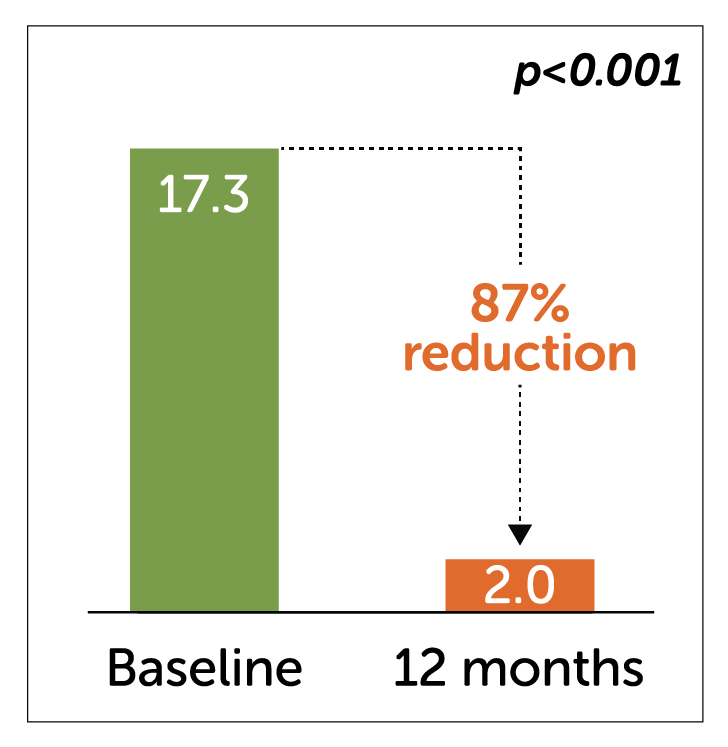
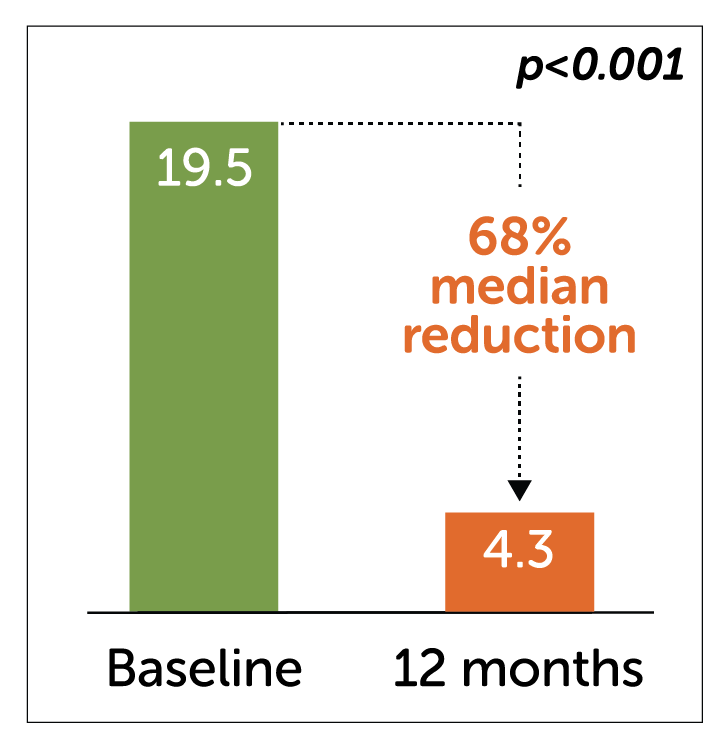
Median Weekly Vomiting Frequency2-3
Idiopathic
Diabetic
Proven clinical impact
Enterra Therapy has been studied extensively with long-term follow-up:
- Clinical trials have demonstrated statistically significant improvements in chronic nausea and vomiting2-3
- Durable symptom improvement observed at 1-, 5-, and 10-year follow-up in numerous, randomized, controlled trials2-5
- At 5 years, 80% of patients treated with Enterra Therapy reported satisfaction with symptom relief4,8
High patient demand
Over 700,000 patients in the U.S. are estimated to have gastroparesis6,*
Over 70,000 patients nationwide have registered on our website to learn more about Enterra Therapy7
In a survey of 1,423 patients with gastroparesis,
only 4% reported satisfaction with available therapies8
Established reimbursement
Enterra Therapy has established reimbursement pathways and is covered by many insurance plans. The Medicare national average payment for a new system implant—including the neurostimulator (CPT 64590) and two unipolar leads (CPT 43647)—is reported as $19,820 as a packaged rate for hospital outpatient procedures, and $16,244 (neurostimulator) plus $9,997 (leads) in the ASC setting.1 Programs that incorporate Enterra Therapy may evaluate these reimbursement structures as part of broader considerations related to patient access. Reimbursement will vary depending on the site of care.†
Interested in offering Enterra Therapy?
Take the next step in expanding treatment options.
Request a conversation with your local Enterra Therapy consultant.
Enterra Medical is proud to provide support to help launch a gastroparesis program, including:
- Medical education and training (optional)
- Procedural and clinical resources
- Ongoing program support (as needed)
*It is likely that a small subset of these patients are potential candidates for Enterra Therapy after considering gastric emptying tests, etiology, medication use, and patient candidacy.
†Reimbursement information provided by Enterra Medical is for general guidance only and does not guarantee coverage or payment. Coding, coverage, and payment policies vary by payer, plan, and geographic region and are subject to change. It is the responsibility of each provider and facility to verify coverage criteria, select appropriate codes, and determine medical necessity for all services rendered. Enterra Medical does not provide legal or billing advice and makes no representation or warranty regarding the completeness or accuracy of the information. All final billing decisions should be made by the treating provider in consultation with the applicable payer.
- Centers for Medicare and Medicaid Services. CMS-1834-FC: Hospital Outpatient Prospective Payment and Ambulatory Surgical Center Payment Systems and Quality Reporting Programs. CY 2026 NFRM ASC Addendum.
- McCallum RW, Snape W, Brody F, Wo J, Parkman HP, Nowak T. Gastric electrical stimulation with Enterra therapy improves symptoms from diabetic gastroparesis in a prospective study. Clin Gastroenterol Hepatol. 2010;8(11):947-e116. doi:10.1016/j.cgh.2010.05.020.
- McCallum RW, Snape W, Brody F, Wo J, Parkman HP, Nowak T. Gastric electrical stimulation with Enterra therapy improves symptoms of idiopathic gastroparesis. Neurogastroenterol Motil (2013) 25, 815–e636. *Manufacture sponsored study. Cross-over periods showed a non-significant reduction in vomiting in the ON vs. OFF period.
- Cassidy DJ, Gerull W, Zike VM, Awad MM. Clinical outcomes of a large, prospective series of gastric electrical stimulation patients using a multidisciplinary protocol. Surg Endosc. 2024.
- Hedjoudje A, Huet E, Leroi AM, Desprez C, Melchior C, Gourcerol G. Efficacy of gastric electrical stimulation in intractable nausea and vomiting at 10 years: A retrospective analysis of prospectively collected data. Neurogastroenterol Motil. 2020;32(11):e13949. doi:10.1111/nmo.13949.
- Ye Y, Yin Y, Huh SY, Almansa C, Bennett D, Camilleri M. Epidemiology, Etiology, and Treatment of gastroparesis: Real-World Evidence From a Large US National Claims Database. Gastroenterology. 2022;162(1):109-121. doi:10.1053/j.gastro.2021.09.064. | A subset of these patients will be eligible for Enterra Therapy.
- Internal Patient opt-in database.
- Yu D, Ramsey FV, Norton WF, et al. The Burdens, Concerns, and Quality of Life of Patients with gastroparesis. Dig Dis Sci. 2017;62(4):879-893.
IMPORTANT SAFETY INFORMATION
Enterra Therapy for treatment of chronic, resistant to medication nausea and vomiting associated with gastroparesis caused by diabetes or an unknown origin in patients aged 18 to 70 years: patients should always discuss potential risks and benefits of the device with their physician.
HUMANITARIAN DEVICE
Authorized by Federal law for use in the treatment of chronic intractable (drug refractory) nausea and vomiting secondary to gastroparesis of diabetic or idiopathic etiology in patients aged 18 to 70 years. The effectiveness of this device for this use has not been demonstrated. What does this mean?